Our scientific board
Technological breakthroughs cannot be achieved without a strong foundation in clinical reality. That is why our scientific board plays an operational, critical, and ongoing role throughout the development of our innovations.
Our 4 recommendations
Our exclusive technology AnCor® has been designed to adapt to a variety of demanding medical uses, from daily care to advanced surgery. Thanks to its unique anchoring and rapid light-curing, our technology opens up new therapeutic avenues in four key areas.
Everyday bandages new generation
Our first consumer product is based on the instantaneous photopolymerization liquid adhesive directly onto the wound (cracks, cuts, etc.).
Unlike conventional liquid films, AnCor® technology allows the formation of a waterproof, flexible, and durable bandage for 48 hours, even underwater.
Fewer applications, better protection, enhanced comfort.
Waterproofing surgical sutures (Surgical Sealant)
AnCor® Tech enables Strong, fast, and secure closure of surgical sutures (organs, mucous membranes, blood vessels) to prevent leaks and thus post-operative complications.
Today, our teams’ priority is to develop a product dedicated to’sealing of lung sutures.
Skin suture
No adhesive currently on the market can effectively close skin wounds. AnCor® technology is behind the development of first device capable of replacing sutures to close the skin in an atraumatic manner.
5 times faster Compared to traditional suturing, this procedure reduces manipulation and preserves skin integrity, resulting in a significantly superior aesthetic result according to our preclinical data.
Treatment of chronic wounds (Advanced Wound Care)
Designed for chronic wounds and acute wounds that cannot be sutured, our Advanced Wound Care research project aims to develop a single-use dressing that can be left in place until the wound has healed.
The goal is to form a stable adhesive interface which promotes healing and protects weakened tissue over several weeks.
One platform, multiple use cases
Thanks to the modularity of its formulation and its proven effectiveness on various biological tissues, AnCor® technology is positioned as a unique foundation for developing a range of innovative medical devices.
These devices are designed to meet the needs of modern medicine: faster, safer, more respectful of living things.
One innovation ready to transform daily skincare
Leveraging our AnCor® technology, we are currently developing an over-the-counter (OTC) medical device., Designed to meet the needs of everyday skin healing: cuts, cracks, small wounds.
This product is based on a single light-curing formulation, enabling quick application, long-lasting adhesion, and effective protection against moisture and external aggressions.
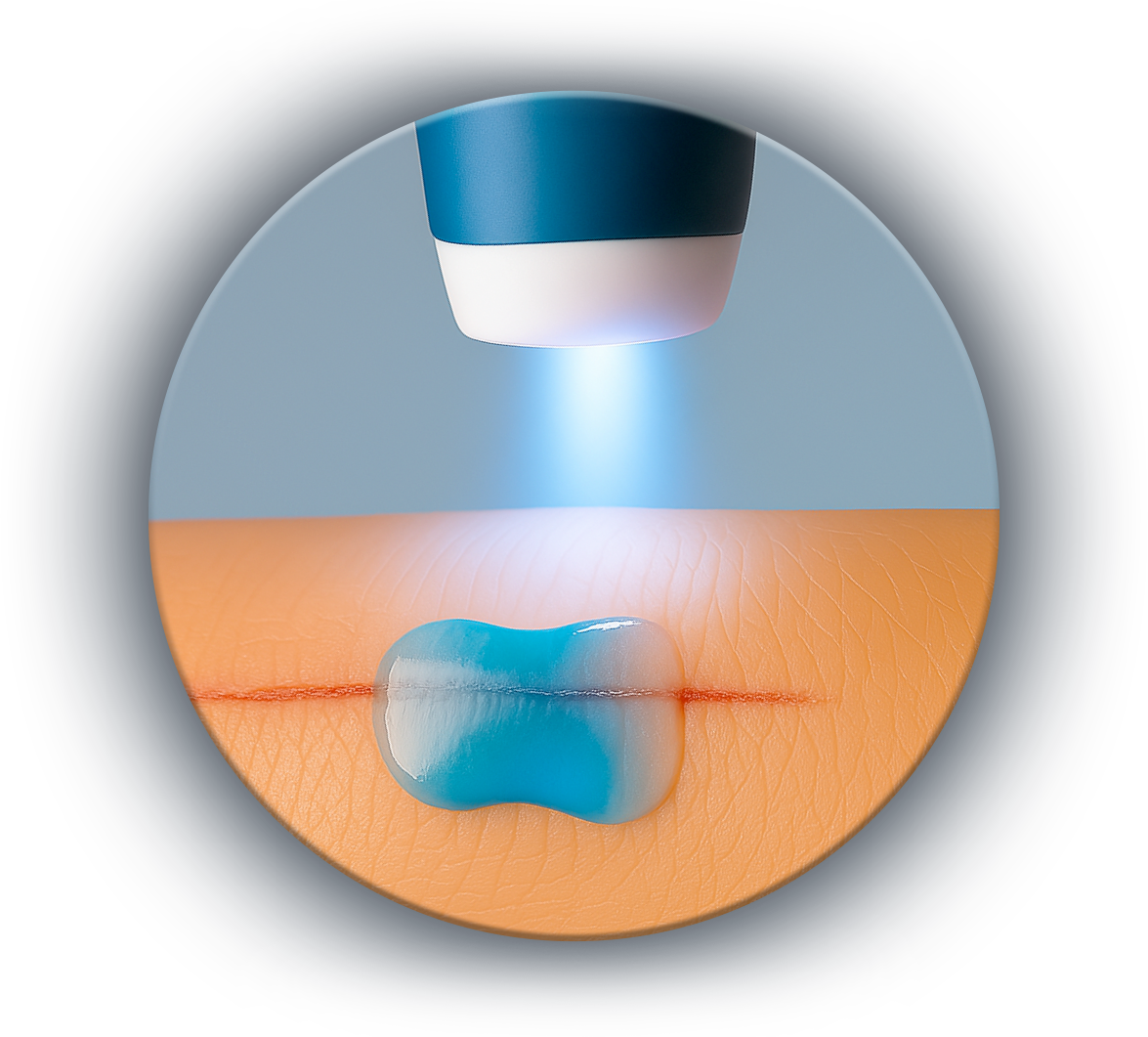
The first product developed by AnCor® is an OTC product that will go on sale in 2026 in the United States and in 2028 in Europe and Asia.
COHESIVES has signed an exclusive license agreement for the AnCor® technology patents with a major player in the international OTC market. The industrial partner is responsible for finalizing the development and marketing of the product.
This first OTC product marks the beginning of a wider rollout of AnCor® technology to the general public. It embodies our desire to democratize access to more effective, faster, and better-tolerated wound healing, thanks to a breakthrough innovation from French medtech research.
Revolutionizing the sealing of internal sutures
The waterproofing of surgical sutures is a challenge.
We have decided to make this our spearhead.
At COHESIVES, we firmly believe that the future of surgery lies in devices that are tailored to specific needs, more effective, and easier to use. It is with this in mind that we have developed AnCor® Tech, a breakthrough innovation in the field of internal tissue adhesion and healing.
Exclusive technology, designed for living tissue
Unlike conventional surgical adhesives, which are often ineffective on wet tissue, AnCor® anchors itself to the surface of biological tissue, penetrating to a depth of 50 to 100 microns to create a strong bond.
AnCor® does not cover, it anchors.
It is resistant to movement, moisture, and physiological stress.
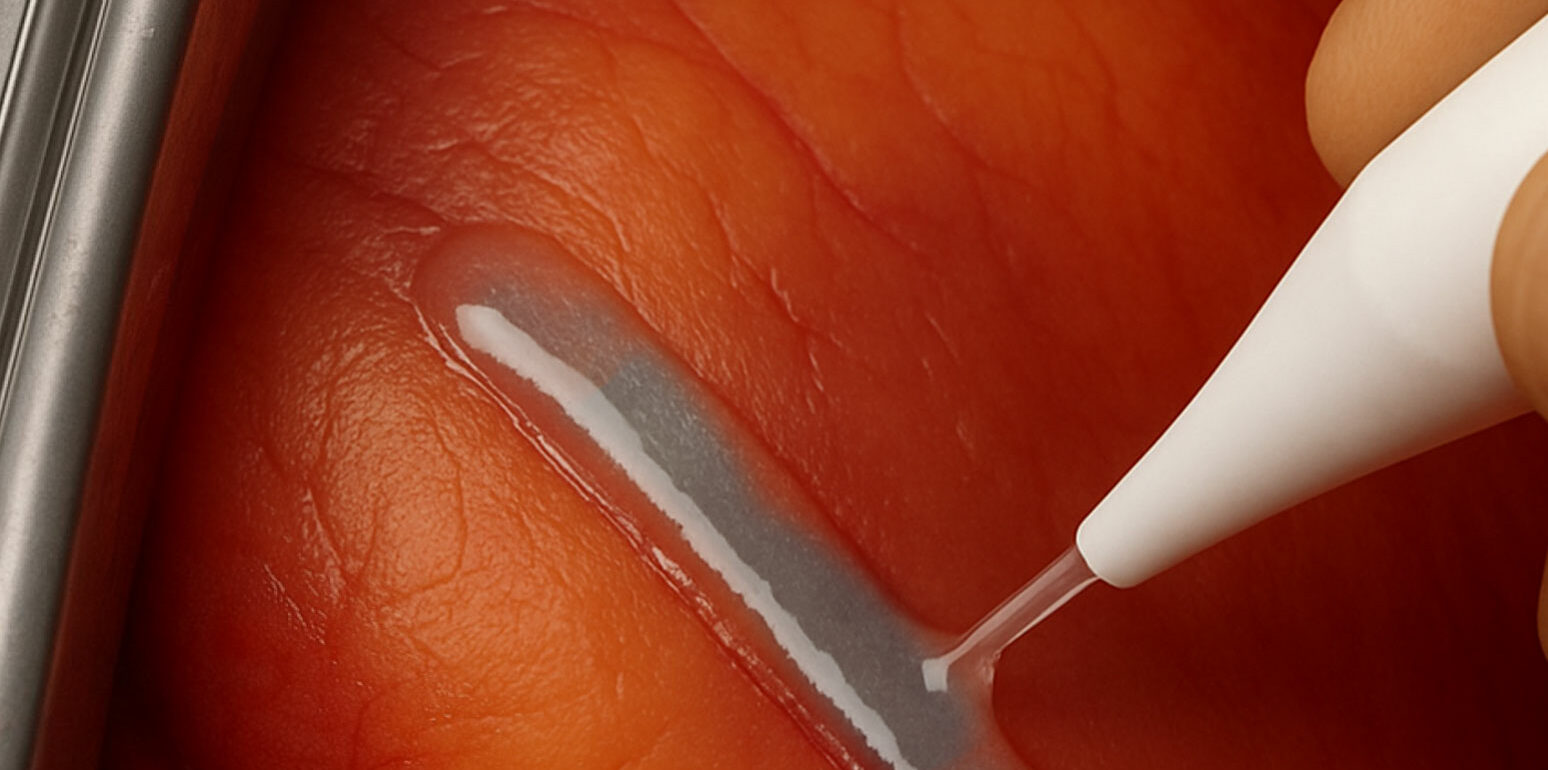
One immediate sealing, optimized healing
Thanks to its light-curing formula, our adhesive transforms into a strong, waterproof polymer film in just 60 seconds under UV light.
A thin adhesive layer is applied to the sutured incision to seal it, then a second thin adhesive layer is applied to seal the entire area. The surgical suture is sealed and protected, giving the surgeon greater confidence.

A long-awaited breakthrough innovation, a concrete response
Sealing internal sutures is one of the most critical and poorly covered areas of modern surgery.
With AnCor®, COHESIVES finally provides a reliable, fast, and reproducible solution that is tailored to the realities of the operating room.
It's more than just an adhesive.
It is a new way of closing tissue with precision, respect, and efficiency.
Our product Novacika®
Novacika®: our clinical evidence on living organisms, demanding and in motion
Even before entering the operating room, our technology has proven itself... in the most demanding healthcare settings.
Novacika®, the first product developed and marketed by COHESIVES, is a synthetic spray-on skin designed for the treatment of traumatic wounds healing by secondary intention in horses.
Far from being a simple veterinary detour, this product is a strategic milestone:
It demonstrates the robustness of our AnCor® technology in complex biological conditions.
It embodies a real-world application that has been clinically validated and appreciated by practitioners.
One single solution,
born to replace skin
Novacika® temporarily replaces the epidermis, protecting the wound throughout the tissue regeneration process. Novacika® gradually falls off as the natural epidermis is restored.
Once light-cured, the adhesive creates a synthetic adhesive film that:
- Stabilizes the wound
- Prevents hypergranulation
- Stops exudate flow and protects the wound from bacteria
- Avoid any immobilization
« The animal can return to the pasture 24-48 hours after application.. »
One proven effectiveness in a real clinical setting
Two studies support its effectiveness:
- A randomized prospective study conducted by VetAgro Sup
- The HOW study, a multicenter study conducted under real-world conditions, including more than 50 cases treated by 8 veterinarians
Key results
No infection
No infection or hypergranulation observed
35 days
average healing time without secondary dressing
2 to 3 visits
for comprehensive care without the use of sedation
A strategic stepping stone to human medicine
Horses are not mere models: they are powerful, fragile, mobile patients—exactly what our technology aims to master.
By providing an innovative solution to a complex veterinary problem, COHESIVES demonstrates that its technology is already operational.
